Application Note B-TA1077
Introduction
Limestone is a representative mineral resource containing calcium carbonate (CaCO₃) as its main component and is widely used in the mining and mineral processing industries as a raw material for producing calcium oxide (CaO) through a calcination process. Limestone undergoes decarboxylation upon heating, release carbon dioxide, so that the reaction temperature and amount are important indicators in ore quality evaluation and process design.
In this application, the thermal decomposition behavior of calcium carbonate in limestone was evaluated using TG-DSC, which simultaneously performs thermogravimetric analysis (TG) and differential scanning calorimetry (DSC).
Measurement and analysis example
Calcium carbonate powder derived from limestone was used as the sample. The sample mass was set to 10 mg, and the measurement was carried out under an air flow atmosphere of 200 mL/min at a heating rate of 10 °C/min from room temperature up to 850 °C. TG-DSC measurements simultaneously recorded the mass change and the endothermic/exothermic behavior of the sample.
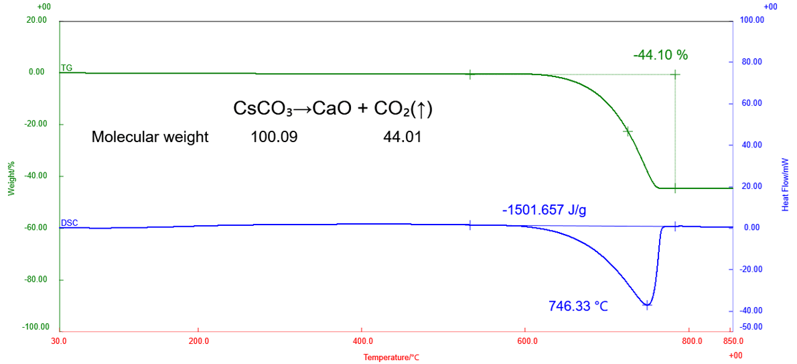
Figure 1: TG-DSC Measurement results
In the TG curve, a monotonic mass loss was observed starting at around 550 °C, and an accompanying endothermic peak was detected in the DSC curve in the same temperature range.
This behavior corresponds to the reaction in which calcium carbonate in limestone undergoes decarbonation, resulting in the formation of calcium oxide accompanied by the release of carbon dioxide.
CaCO₃ → CaO + CO₂↑
The mass loss observed in the TG curve was approximately 44%, which agrees well with the stoichiometric mass loss calculated from the molecular weight of calcium carbonate.
These results confirm that, under the present measurement conditions, calcium carbonate in the limestone undergoes an almost complete decarbonation. In addition, the reaction energy associated with the decarbonation was estimated to be approximately 1500 J/g from the area of the endothermic peak in the DSC curve.
TG-DSC measurements demonstrated that the decarbonation of calcium carbonate in limestone can be quantitatively evaluated in terms of both mass change and reaction energy.
This method is considered effective for quality evaluation of limestone, investigation of calcination conditions, and process design in the mining and mineral processing fields.

